Research:
Principle of Differential Sectioning Microscopy
To understand the principle of differential sectioning microscopy, it is necessary to have some basic knowledge of optical sectioning microscopy. Confocal microscopy is the most popular sectioning microscopy, which provides three-dimensional images of micro-objects and living cells. An excellent reference book is Confocal Microscopy by T. Wilson (Academic Press, London, 1990).
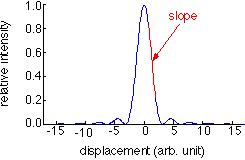
In conventional confocal microscopy, the signal light comes mainly from the focal region. Light from outside of the focal region is heavily attenuated by a pinhole in front of the detector. The axial response curve is shown as the figure above, where the amount of light that enters the detector is plotted as a function of sample position. The optical sectioning ability is usually defined as the width of the axial response curve, d.
Although in confocal microscopy one cannot resolve two regions of height separation smaller than d, there are many applications in which light comes from a region much narrower than d. One example is when the signal light comes from specular reflection or fluorescence from samples with well defined surfaces, such as cell membranes. In such cases the resolution is determined by one's ability to locate the position from which the signal comes, instead of the width of the confocal response curve. One can see from the figure that placing the sample exactly at the focal point is not the most advantageous for achieving high depth resolution; the zero derivative of the response curve with respect to the sample position means least sensitivity to sample height variation. In contrast, if one places the sample slightly away from the focal point, so that its position is at the slopes of the response curve, the sensitivity becomes greatest. At the slopes, sample height variation causes a differential change of signal. The sensitivity of this effect can be utilized to image surface structures with great depth resolution. We first demonstrated this principle using confocal microscopy, and got the patent of it (US Patent No. 5804813; Taiwan Patent No. 082212).
The above idea is applicable to other kinds of sectioning microscopy. For example, one may use structured illumination to produce widefield sectioning microscopy. With the setup of widefield sectioning microscopy, our differential sectioning microscopy works on a conventional optical microscope, and thus is easy to setup and deploy. We now call this technique "non-interferometric widefield optical profilometry," or its acronym, NIWOP.
Resolution and Dynamic Range of Differential Sectioning Microscopy
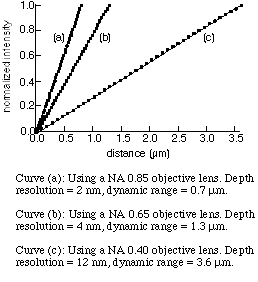
The linearity and dynamic range of differential confocal microscopy with respective to objective lenses of different numerical apertures are shown as the figure below. The depth resolution is limited by the system noise, including power fluctuation of the light source, shot and thermal noise from signal light and photodetectors, as well as quantization error of the data acquisition system. For reflective signals, the dominant noise usually comes from power fluctuation of the light source.
Using a laser with 0.2% power fluctuation as the light source, the uncertainty of curve (a) in this figure corresponds to 2-nm depth resolution. The dynamic range of curve (a) is 0.7 micrometers. For a larger dynamic range, one can use an objective lens with lower numerical aperture. The dynamic range of curve (c) is as large as 3.6 micrometers. However, the depth resolution is 12 nm.
Sample Images
Here are some images obtained with our differential confocal microscope. They are measured with 633-nm light.
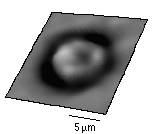
The shape of normal red blood cells is known to be like a disc with the center thinner than the edge. The mean value of the recess in the center is 700 nm and the standard deviation is 150 nm. This image shows a 3-dimensional image of a human red blood cell under differential confocal microscope. The measurement was taken with a N.A.=0.85 objective lens. The center recess we measured is 570 nm. The dark ring around the edge of the cell is due to the steep slope of the surface, where little light is reflected. Although images of metal-coated red blood cells (or other biological tissues) under scanning electron microscope can be found in textbooks, this image demonstrates that we can observe fresh untreated biological samples under nature environment with great depth resolution.
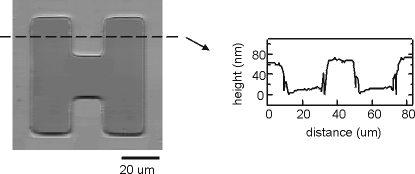
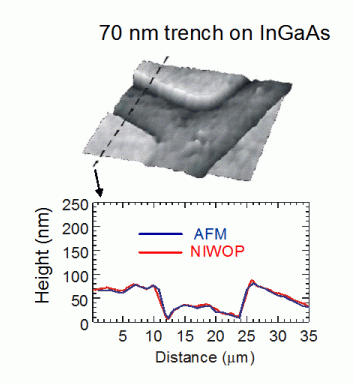
This is the image and profile of an H-shape trench on InGaAs. The depth of this trench is only 70 nm. From the profile we see that differential confocal microscopy can reveal profile details down to a few nanometers.
This is an image of a layer of collagen fibers extracted from bovine skin tissues. The diameters of collagen fiber measured by scanning electron microscopy are about 100 nm, which is apparently smaller than the 0.3-µm lateral resolution of our system. However, because of the high depth sensitivity of differential confocal microscopy, the lateral distribution of the fiber becomes measurable. The lateral resolution could be further improved by using oil-immersed objectives and shorter wavelength light sources.
And here are images from our NIWOP technique.
This is an image of a 70-nm deep trench on InGaAs. We used atomic force microscope to profile the same area and obtained very close results.
Because the NIWOP system has imaging speed much higher than the confocal system, we employ it for membrane studies on living cells.
Thin-film characterizations
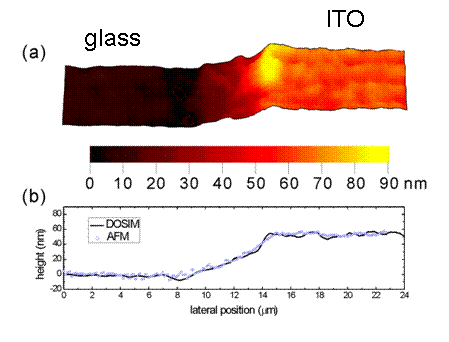
In the NIWOP system, if we use a narrow-band light source to illuminate a transparent thin film, the Fabry-Perot interferometric effect will produce the phase information which is determined by the refractive index and thickness of this thin film. Therefore we can calculate the refractive index and thickness from the measured image intensity. We call this technique "differential optical sectioning interference microscopy," or its acronym DOSIM. Here is an image obtained by DOSIM:
In this figure, the sample is an ITO film coated on glass. The profile obtained by DOSIM is very close to that obtained by AFM. We also obtained that the refractive index of the ITO is 1.79 ± 0.018. The average index on a large area of the same specimen measured by optical ellipsometry is 1.816. This technique provides a method to obtain both the refractive index and thickness of a thin film with a simple setup. This technique obtains the patent in the US (US Patent No. 7545510).
Publications about Differential Sectioning Microscopy
1. Chau-Hwang Lee and Jyhpyng Wang, "Noninterferometric differential confocal microscopy with 2-nm depth resolution," Optics Communications 135, 233 (1997).
2. Chao-Wei Tsai, Chau-Hwang Lee, and Jyhpyng Wang, "Deconvolution of local surface response from topography in nanometer profilometry with a dual-scan method," Optics Letters 24, 1732 (1999).
3. Chau-Hwang Lee, Hong-Yao Mong, and Wan-Chen Lin, "Non-interferometric wide-field optical profilometry with nanometer depth resolution," Optics Letters 27, 1773 (2002).
4. Chun-Chieh Wang, Jiunn-Yuan Lin, Hung-Jhang Jian, and Chau-Hwang Lee, "Transparent thin-film characterization by using differential optical sectioning interference microscopy," Applied Optics 46, 7460 (2007).